Page 12 - Flipbook
P. 12
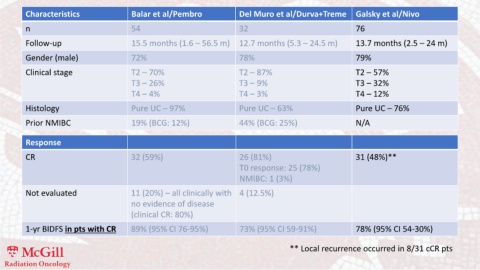
Characteristics Balar et al/Pembro Del Muro et al/Durva+Treme Galsky et al/Nivo
n 54 32 76
Follow-up 15.5 months (1.6 – 56.5 m) 12.7 months (5.3 – 24.5 m) 13.7 months (2.5 – 24 m)
Gender (male) 72% 78% 79%
Clinical stage T2 – 70% T2 – 87% T2 – 57%
T3 – 26% T3 – 9% T3 – 32%
T4 – 4% T4 – 3% T4 – 12%
Histology Pure UC – 97% Pure UC – 63% Pure UC – 76%
Prior NMIBC 19% (BCG: 12%) 44% (BCG: 25%) N/A
Response
CR 32 (59%) 26 (81%) 31 (48%)**
T0 response: 25 (78%)
NMIBC: 1 (3%)
Not evaluated 11 (20%) – all clinically with 4 (12.5%)
no evidence of disease
(clinical CR: 80%)
1-yr BIDFS in pts with CR 89% (95% CI 76-95%) 73% (95% CI 59-91%) 78% (95% CI 54-30%)
** Local recurrence occurred in 8/31 cCR pts
Radiation Oncology