Page 1 - DDI Clinical Tool
P. 1
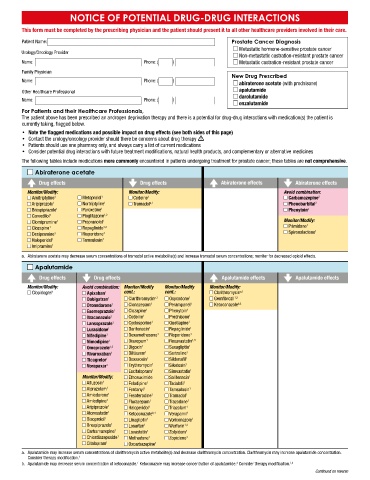
NOTICE OF POTENTIAL DRUG-DRUG INTERACTIONS
This form must be completed by the prescribing physician and the patient should present it to all other healthcare providers involved in their care.
Patient Name: Prostate Cancer Diagnosis
Metastatic hormone-sensitive prostate cancer
Urology/Oncology Provider
Non-metastatic castration-resistant prostate cancer
Name: Phone: ( ) Metastatic castration-resistant prostate cancer
Family Physician
New Drug Prescribed
Name: Phone: ( )
abiraterone acetate (with prednisone)
Other Healthcare Professional apalutamide
darolutamide
Name: Phone: ( )
enzalutamide
For Patients and their Healthcare Professionals,
The patient above has been prescribed an androgen deprivation therapy and there is a potential for drug-drug interactions with medication(s) the patient is
currently taking, flagged below.
• Note the flagged medications and possible impact on drug effects (see both sides of this page)
• Contact the urology/oncology provider should there be concerns about drug therapy m
• Patients should use one pharmacy only, and always carry a list of current medications
• Consider potential drug interactions with future treatment modifications, natural health products, and complementary or alternative medicines
The following tables include medications more commonly encountered in patients undergoing treatment for prostate cancer; these tables are not comprehensive.
Abiraterone acetate
Drug effects Drug effects Abiraterone effects Abiraterone effects
Monitor/Modify: Monitor/Modify: Avoid combination:
Amitriptyline 1 Metoprolol 1 Codeine 1 Carbamazepine 2
Aripiprazole 1 Nortriptyline 1 Tramadol a,1 Phenobarbital 2
Brexpiprazole 1 Paroxetine 1 Phenytoin 2
Carvedilol 1 Pioglitazone 1,2
Clomipramine 1 Propranolol 1 Monitor/Modify:
1
Clozapine 1 Repaglinide 1,2 Primidone 1
Desipramine 1 Risperidone 1 Spironolactone
Haloperidol 1 Tamsulosin 1
Imipramine 1
a. Abiraterone acetate may decrease serum concentrations of tramadol active metabolite(s) and increase tramadol serum concentrations; monitor for decreased opioid effects.
Apalutamide
Drug effects Drug effects Apalutamide effects Apalutamide effects
Monitor/Modify: Avoid combination: Monitor/Modify Monitor/Modify Monitor/Modify:
Clopidogrel 1 Apixaban 1 cont.: cont.: Clarithromycin a,1
Dabigatran 1 Clarithromycin a,1 Oxycodone 1 Gemfibrozil 1,3
Dronedarone 1 Clonazepam 1 Perampanel 1 Ketoconazole b,3
Esomeprazole 1 Clozapine 1 Phenytoin 1
Itraconazole 1 Codeine 1 Prednisone 1
Lansoprazole 1 Cyclosporine 1 Quetiapine 1
Lurasidone 1 Darifenacin 1 Repaglinide 1
Nifedipine 1 Dexamethasone 1 Risperidone 1
Nimodipine 1 Diazepam 1 Rosuvastatin 1,3
Omeprazole 1,3 Digoxin 1 Saxagliptin 1
Rivaroxaban 1 Diltiazem 1 Sertraline 1
Ticagrelor 1 Doxazosin 1 Sildenafil 1
Vorapaxar 1 Erythromycin 1 Silodosin 1
Escitalopram 1 Simvastatin 1
Monitor/Modify: Ethosuximide Solifenacin 1
Alfuzosin 1 Felodipine 1 Tadalafil 1
Alprazolam 1 Fentanyl 1 Tamsulosin 1
Amiodarone 1 Fesoterodine 1 Tramadol 1
Amlodipine 1 Flurazepam 1 Trazodone 1
Aripiprazole 1 Haloperidol 1 Triazolam 1
Atorvastatin 1 Ketoconazole b,1 Verapamil 1
Bisoprolol 1 Linagliptin 1 Voriconazole 1
Brexpiprazole 1 Losartan 1 Warfarin 1,3
Carbamazepine 1 Lovastatin 1 Zolpidem 1
Chlordiazepoxide 1 Methadone 1 Zopiclone 1
Citalopram 1 Oxcarbazepine 1
a. Apalutamide may increase serum concentrations of clarithromycin active metabolite(s) and decrease clarithromycin concentration. Clarithromycin may increase apalutamide concentration.
Consider therapy modification. 1
b. Apalutamide may decrease serum concentration of ketoconazole. Ketoconazole may increase concentration of apalutamide. Consider therapy modification.
1,3
1
3
Continued on reverse