Page 12 - CUA 2020_Onco_Prostate
P. 12
2020 CUA Abstracts
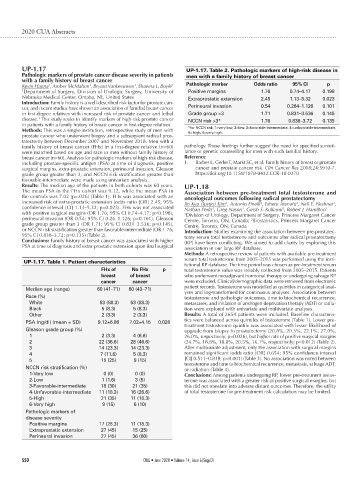
UP-1.17 UP-1.17. Table 2. Pathologic markers of high-risk disease in
Pathologic markers of prostate cancer disease severity in patients men with a family history of breast cancer
with a family history of breast cancer
1
1
Kevin Hanna , Amber McMahon , Bryant VanLeeuwen , Shawna L. Boyle 1 Pathologic marker Odds ratio 95% CI p
1
1 Department of Surgery, Division of Urologic Surgery, University of Positive margins 1.76 0.74–4.17 0.198
Nebraska Medical Center, Omaha, NE, United States Extraprostatic extension 2.45 1.13–5.32 0.023
Introduction: Family history is a well-described risk factor for prostate can-
cer, and recent studies have shown an association of familial breast cancer Perineural invasion 0.54 0.264–1.126 0.101
in first-degree relatives with increased risk of prostate cancer and lethal Grade group >3 1.71 0.831–3.536 0.145
1
disease. This study seeks to identify markers of high-risk prostate cancer NCCN risk >3* 1.76 0.838–3.72 0.135
in patients with a family history of breast cancer in first-degree relatives. *for NCCN risk, 1=very low, 2=low, 3=favorable-intermediate, 4=unfavorable-intermediate,
Methods: This was a single-institution, retrospective study of men with 5= high, 6=very high.
prostate cancer who underwent biopsy and a subsequent radical pros-
tatectomy between December 2007 and November 2018. Men with a
family history of breast cancer (FHx) in a first-degree relative (n=60) pathology. These findings further suggest the need for specified surveil-
were matched based on age and race to men without family history of lance or genetic counselling for men with such familial history.
breast cancer (n=60). Analysis for pathologic markers of high-risk disease, Reference
including prostate-specific antigen (PSA) at time of diagnosis, positive 1. Barber L, Gerke T, Markt SC, et al. Family history of breast or prostate
surgical margins, extra-prostatic extension, perineural invasion, Gleason cancer and prostate cancer risk. Clin Cancer Res 2018;24:5910-7.
grade group greater than 3, and NCCN risk stratification greater than https://doi.org/10.1158/1078-0432.CCR-18-0370
favorable-intermediate were made using univariate analysis.
Results: The median age of the patients in both cohorts was 60 years. UP-1.18
The mean PSA in the FHx cohort was 9.12, while the mean PSA in Association between pre-treatment total testosterone and
the controls was 7.02 (p=.026) (Table 1). FHx was associated with an oncological outcomes following radical prostatectomy
increased risk of extra-prostatic extension (odds ratio [OR] 2.45; 95% Jin Kyu (Justin) Kim , Antonio Finelli , Eshetu Atenafu , Neil E. Fleshner ,
2
1
1
1
confidence interval [CI] 1.13–5.32; p=0.023). FHx was not associated Nathan Perlis , Greg Nason , Girish S. Kulkarni , Robert J. Hamilton 1
1
1
1
with positive surgical margins (OR 1.76; 95% CI 0.74–4.17; p=0.198), 1 Division of Urology, Department of Surgery, Princess Margaret Cancer
perineural invasion (OR 0.54; 95% CI 0.26–1.126; p=0.101), Gleason Centre, Toronto, ON, Canada; Biostatistics, Princess Margaret Cancer
2
grade group greater than 3 (OR 1.71; 95% CI 0.831–3.536; p=0.145), Centre, Toronto, ON, Canada
or NCCN risk stratification greater than favorable-intermediate (OR 1.76; Introduction: Studies examining the association between pre-prostatec-
95% CI 0.838–3.72; p=0.135) (Table 2). tomy serum total testosterone and outcomes after radical prostatectomy
Conclusions: Family history of breast cancer was associated with higher (RP) have been conflicting. We aimed to add clarity by exploring this
PSA at time of diagnosis and extra-prostatic extension upon final surgical association in our large RP database.
Methods: A retrospective review of patients with available pre-treatment
serum total testosterone from 2005–2015 was performed using the insti-
UP-1.17. Table 1. Patient characteristics tutional RP database. The time period was chosen as pre-treatment serum
FHx of No FHx p total testosterone value was reliably collected from 2005–2015. Patients
breast of breast who underwent neoadjuvant hormonal therapy or undergoing salvage RP
cancer cancer were excluded. Clinical/demographic data were retrieved from electronic
patient records. Testosterone was modelled as quintiles in categorical anal-
Median age (range) 60 (41–71) 60 (43–71)
yses and log-transformed for continuous analyses. Association between
Race (%) testosterone and pathologic outcomes, time to biochemical recurrence,
White 53 (88.3) 53 (88.3) metastases, and initiation of androgen deprivation therapy (ADT) or radia-
Black 5 (8.3) 5 (8.3) tion were explored with univariate and multivariate analyses.
Other 2 (3.3) 2 (3.3) Results: A total of 2654 patients were included. Baseline characteris-
tics were balanced across quintiles of testosterone (Table 1). Lower pre-
PSA (ng/dl) (mean ± SD) 9.12±6.86 7.02±4.15 0.026
treatment testosterone quintile was associated with lesser likelihood of
Gleason grade group (%) upgrade from biopsy to prostatectomy (20.8%, 20.3%, 22.1%, 27.0%,
1 2 (3.3) 4 (6.6) 26.0%, respectively; p=0.026), but higher rate of positive surgical margins
2 22 (36.6) 28 (46.6) (24.7%, 18.8%, 18.8%, 20.5%, 16.1%, respectively; p=0.012) (Table 2).
3 14 (23.3) 14 (23.3) After multivariate adjustment, only the association with surgical margins
4 7 (11.6) 5 (8.3) remained significant (odds ratio [OR] 0.654; 95% confidence interval
5 15 (25) 9 (15) [CI] 0.511–0.838; p<0.001) (Table 3). No association was noted between
testosterone and time to biochemical recurrence, metastasis, salvage ADT,
NCCN risk stratification (%) or radiation (Table 4).
1-Very low 0 (0) 0 (0) Conclusions: Among patients undergoing RP, lower pre-treatment testos-
2-Low 1 (1.6) 3 (5) terone was associated with a greater risk of positive surgical margins, but
3-Favorable-intermediate 18 (30) 21 (35) this did not translate into adverse distant outcomes. Therefore, the utility
4-Unfavorable-intermediate 11 (18.3) 16 (26.6) of total testosterone for pre-treatment risk calculation may be limited.
5-High 21 (35) 11 (18.3)
6-Very high 9 (15) 6 (10)
Pathologic markers of
disease severity
Positive margins 17 (28.3) 11 (18.3)
Extraprostatic extension 27 (45) 15 (25)
Perineural invasion 27 (45) 36 (60)
S50 CUAJ • June 2020 • Volume 14, Issue 6(Suppl2)