Page 2 - CUA ESMO ToolCard WEB
P. 2
SELECT EVIDENCE
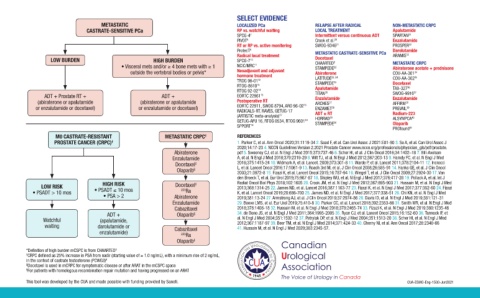
METASTATIC LOCALIZED PCa RELAPSE AFTER RADICAL NON-METASTATIC CRPC
CASTRATE-SENSITIVE PCa RP vs. watchful waiting LOCAL TREATMENT Apalutamide
SPCG-4 7 Intermittent versus continuous ADT SPARTAN 31
PIVOT 8 Crook et al. 20 Enzalutamide
RT or RP vs. active monitoring SWOG-9346 21 PROSPER 32
ProtecT 9 Darolutamide
Radical local treatment METASTATIC CASTRATE-SENSITIVE PCa ARAMIS 33
LOW BURDEN HIGH BURDEN SPCG-7 10 Docetaxel 5 METASTATIC CRPC
CHAARTED
• Visceral mets and/or ≥ 4 bone mets with ≥ 1 NCIC/MRC 11 STAMPEDE 22 Abiraterone acetate + prednisone
outside the vertebral bodies or pelvis* Neoadjuvant and adjuvant Abiraterone COU-AA-301 34
hormone treatment 23, 24 35
TROG 96-01 12 LATITUDE 25 COU-AA-302
RTOG-8610 13 STAMPEDE Docetaxel 36
RTOG 92-02 14 Apalutamide TAX-327 37
26
ADT + Prostate RT + ADT + EORTC 22961 15 TITAN SWOG-9916
Enzalutamide
Enzalutamide
(abiraterone or apalutamide (abiraterone or apalutamide Postoperative RT ARCHES AFFIRM 38
27
or enzalutamide or docetaxel) or enzalutamide or docetaxel) EORTC 22911, SWOG 8794, ARO 96-02 16 ENZAMET 28 PREVAIL 39
RADICALS-RT, RAVES, GETUG-17
(ARTISTIC meta-analysis) 17 ADT ± RT 29 Radium-223
40
GETUG-AFU 16, RTOG 0534, RTOG 9601 18 HORRAD 30 ALSYMPCA
SPPORT 19 STAMPEDE Olaparib
PROfound 41
M0 CASTRATE-RESISTANT METASTATIC CRPC † REFERENCES
PROSTATE CANCER (CRPC) † 1. Parker C, et al. Ann Oncol 2020;31:1119-34 2. Saad F, et al. Can Urol Assoc J 2021:E81-90 3. So A, et al. Can Urol Assoc J
2020;14:17-23 4. NCCN Guidelines Version 2.2021 Prostate Cancer www.nccn.org/professionals/physician_gls/pdf/prostate.
Abiraterone pdf 5. Sweeney CJ, et al. N Engl J Med 2015;373:737-46 6. Scher HI, et al. J Clin Oncol 2016;34:1402–18 7. Bill-Axelson
Enzalutamide A, et al. N Engl J Med 2018;379:2319-29 8. Wilt TJ, et al. N Engl J Med 2012;367:203-13 9. Hamdy FC, et al. N Engl J Med
Docetaxel ‡ 2016;375:1415-24 10. Widmark A, et al. Lancet. 2009;373:301-8 11. Warde P, et al. Lancet 2011;378:2104-11 12. Incrocci
L, et al. Lancet Oncol 2016;17:1061-9 13. Roach 3rd M, et al. J Clin Oncol 2008;26:585-91 14. Hanks GE, et al. J Clin Oncol
Olaparib § 2003;21:3972-8 15. Fizazi K, et al. Lancet Oncol 2015;16:787-94 16. Wiegel T, et al. J Clin Oncol 2009;27:2924-30 17. Van
den Broeck T, et al. Eur Urol 2019;75:967-87 18. Shipley WU, et al. N Engl J Med 2017;376:417-28 19. Pollack A, et al. Int J
HIGH RISK ‡ Radiat Oncol Biol Phys 2018;102:1605 20. Crook JM, et al. N Engl J Med 2012;367:895-903 21. Hussain M, et al. N Engl J Med
LOW RISK Docetaxel 2013;368:1314-25 22. James ND, et al. Lancet 2016;387:1163-77 23. Fizazi K, et al. N Engl J Med 2017;377:352-60 24. Fizazi
• PSADT > 10 mos • PSADT ≤ 10 mos 223 Ra K, et al. Lancet Oncol 2019;20:686-700 25. James ND, et al. N Engl J Med 2017;377:338-51 26. Chi KN, et al. N Engl J Med
• PSA > 2 Abiraterone 2019;381:13-24 27. Armstrong AJ, et al. J Clin Oncol 2019;37:2974-86 28. Davis ID, et al. N Engl J Med 2019;381:121-31
Enzalutamide 29. Boeve LMS, et al. Eur Urol 2019;75:410-8 30. Parker CC, et al. Lancet 2018;392:2353-66 31. Smith MR, et al. N Engl J Med
Cabazitaxel 2018;378:1408-18 32. Hussain M, et al. N Engl J Med 2018;378:2465-74 33. Fizazi K, et al. N Engl J Med 2019;380:1235-46
ADT + Olaparib § 34. de Bono JS, et al. N Engl J Med 2011;364:1995-2005 35. Ryan CJ, et al. Lancet Oncol 2015;16:152-60 36. Tannock IF, et
Watchful (apalutamide, al. N Engl J Med 2004;351:1502-12 37. Petrylak DP, et al. N Engl J Med 2004;351:1513-20 38. Scher HI, et al. N Engl J Med
2012;367:1187-97 39. Beer TM, et al. N Engl J Med 2014;371:424-33 40. Cherny NI, et al. Ann Oncol 2017;28:2340-66
waiting darolutamide or Cabazitaxel 41. Hussain M, et al. N Engl J Med 2020;383:2345-57.
enzalutamide) 223 Ra
Olaparib ‡
*Definition of high burden mCSPC is from CHAARTED 5
† CRPC defined as 25% increase in PSA from nadir (starting value of ≥ 1.0 ng/mL), with a minimum rise of 2 ng/mL,
in the context of castrate testosterone (PCWG3) 6
‡ Docetaxel is used in mCRPC for symptomatic disease or after ARAT in the mCSPC space
§ For patients with homologous recombination repair mutation and having progressed on an ARAT
This tool was developed by the CUA and made possible with funding provided by Sanofi. CUA-ESMO-Eng-1500-Jun2021