Page 5 - Advanced PC Booklet 17
P. 5
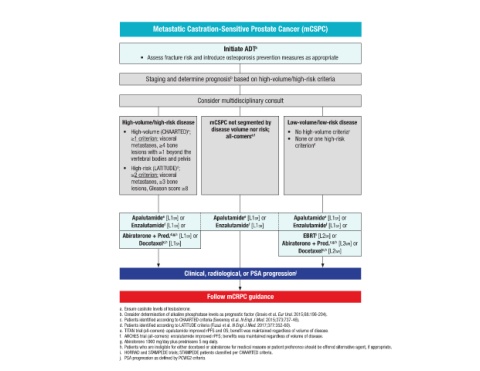
Metastatic Castration-Sensitive Prostate Cancer (mCSPC)
Initiate ADT a
• Assess fracture risk and introduce osteoporosis prevention measures as appropriate
b
Staging and determine prognosis based on high-volume/high-risk criteria
Consider multidisciplinary consult
High-volume/high-risk disease mCSPC not segmented by Low-volume/low-risk disease
• High-volume (CHAARTED) ; disease volume nor risk; • No high-volume criteria c
c
≥1 criterion: visceral all-comers e,f • None or one high-risk
metastases, ≥4 bone criterion d
lesions with ≥1 beyond the
vertebral bodies and pelvis
d
• High-risk (LATITUDE) ;
≥2 criterion: visceral
metastases, ≥3 bone
lesions, Gleason score ≥8
Apalutamide [L1SR] or Apalutamide [L1SR] or Apalutamide [L1SR] or
e
e
e
f
f
f
Enzalutamide [L1SR] or Enzalutamide [L1SR] Enzalutamide [L1SR] or
Abiraterone + Pred. d,g,h [L1SR] or EBRT [L2SR] or
i
Docetaxel [L1SR] Abiraterone + Pred. c,g,h [L3WR] or
c,h
Docetaxel [L2WR]
c,h
Clinical, radiological, or PSA progression j
Follow mCRPC guidance
a. Ensure castrate levels of testosterone.
b. Consider determination of alkaline phosphatase levels as prognostic factor (Gravis et al. Eur Urol. 2015;68:196-204).
c. Patients identified according to CHAARTED criteria (Sweeney et al. N Engl J Med. 2015;373:737-46).
d. Patients identified according to LATITUDE criteria (Fizazi et al. N Engl J Med. 2017;377:352-60).
e. TITAN trial (all-comers): apalutamide improved rPFS and OS; benefit was maintained regardless of volume of disease.
f. ARCHES trial (all-comers): enzalutamide improved rPFS; benefits was maintained regardless of volume of disease.
g. Abiraterone 1000 mg/day plus prednisone 5 mg daily.
h. Patients who are ineligible for either docetaxel or abiraterone for medical reasons or patient preference should be offered alternative agent, if appropriate.
i. HORRAD and STAMPEDE trials; STAMPEDE patients classified per CHAARTED criteria.
j. PSA progression as defined by PCWG2 criteria.