Page 8 - Flipbook
P. 8
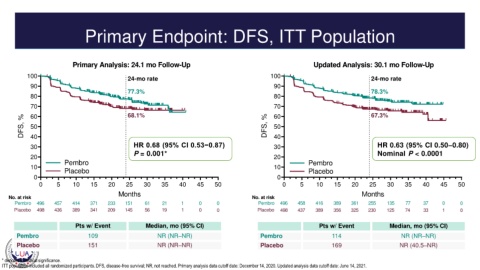
Primary Endpoint: DFS, ITT Population
Primary Analysis: 24.1 mo Follow-Up Updated Analysis: 30.1 mo Follow-Up
100 24-mo rate 100 24-mo rate
90 90
77.3% 78.3%
80 80
70 68.1% 70 67.3%
DFS, % 60 DFS, % 60
50
50
40
40
30 HR 0.68 (95% CI 0.53−0.87) 30 HR 0.63 (95% CI 0.50–0.80)
P = 0.001* Nominal P < 0.0001
20 20
Pembro Pembro
10 10
Placebo Placebo
0 0
0 5 10 15 20 25 30 35 40 45 50 0 5 10 15 20 25 30 35 40 45 50
Months Months
No. at risk No. at risk
Pembro 496 457 414 371 233 151 61 21 1 0 0 Pembro 496 458 416 389 361 255 135 77 37 0 0
Placebo 498 436 389 341 209 145 56 19 1 0 0 Placebo 498 437 389 356 325 230 125 74 33 1 0
Pts w/ Event Median, mo (95% CI) Pts w/ Event Median, mo (95% CI)
Pembro 109 NR (NR–NR) Pembro 114 NR (NR–NR)
Placebo 151 NR (NR–NR) Placebo 169 NR (40.5–NR)
* denotes statistical significance.
ITT population included all randomized participants. DFS, disease-free survival; NR, not reached. Primary analysis data cutoff date: December 14, 2020. Updated analysis data cutoff date: June 14, 2021.