Page 9 - Flipbook
P. 9
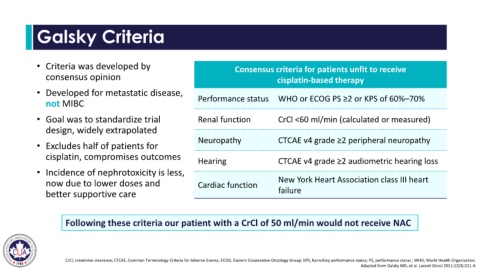
Galsky Criteria
• Criteria was developed by Consensus criteria for patients unfit to receive
consensus opinion cisplatin-based therapy
• Developed for metastatic disease,
not MIBC Performance status WHO or ECOG PS ≥2 or KPS of 60%–70%
• Goal was to standardize trial Renal function CrCl <60 ml/min (calculated or measured)
design, widely extrapolated
Neuropathy CTCAE v4 grade ≥2 peripheral neuropathy
• Excludes half of patients for
cisplatin, compromises outcomes
Hearing CTCAE v4 grade ≥2 audiometric hearing loss
• Incidence of nephrotoxicity is less,
now due to lower doses and Cardiac function New York Heart Association class III heart
better supportive care failure
Following these criteria our patient with a CrCl of 50 ml/min would not receive NAC
CrCl, creatinine clearance; CTCAE, Common Terminology Criteria for Adverse Events; ECOG, Eastern Cooperative Oncology Group; KPS, Karnofsky performance status; PS, performance status ; WHO, World Health Organization.
Adapted from Galsky MD, et al. Lancet Oncol 2011;12(3):211-4.