Page 3 - CUA Adv Prostate Ca Drug Access List-MAR-2022
P. 3
ALBERTA Link to Patient Assistance Programs HOME
Funding:
Cancer patients may receive treatment at cancer centres, their community hospitals, or taken at home.
Alberta provides cancer drugs specified in the Outpatient Cancer Drug Benefit Program, at no charge, to eligible residents for the treatment of cancer.
Formularies:
Alberta Health Drug Benefit List - https://idbl.ab.bluecross.ca/idbl/load.do
Alberta Health Services Outpatient Cancer Drug Benefit Program - https://www.albertahealthservices.ca/assets/programs/ps-1025651-drug-benefit-list.pdf
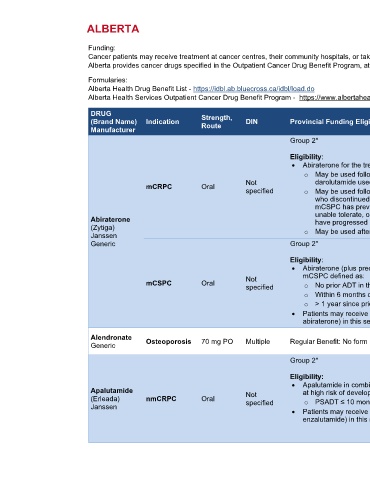
DRUG Strength,
(Brand Name) Indication Route DIN Provincial Funding Eligibility Criteria References
Manufacturer
Group 2* AHS Outpatient
Cancer Drug
Eligibility: Benefit Program
• Abiraterone for the treatment of mCRPC [3-22]
o May be used following progression on or intolerance to apalutamide/enzalutamide/
Not darolutamide used in nmCRPC setting
mCRPC Oral
specified o May be used following apalutamide/enzalutamide used in mCSPC setting for patients
who discontinued due to intolerance or toxicity. Not to be used in patients whose
mCSPC has previously progressed on apalutamide/enzalutamide, unless they are
unable tolerate, or are not candidates for other therapeutic options. Patients must not
Abiraterone have progressed previously on abiraterone.
(Zytiga)
Janssen o May be used after progression on docetaxel if not received before
Generic Group 2* AHS Outpatient
Cancer Drug
Eligibility: Benefit Program
• Abiraterone (plus prednisone) in combination with ADT for treatment of patients with [3-22]
Not mCSPC defined as:
mCSPC Oral o No prior ADT in the metastatic setting, OR
specified
o Within 6 months of beginning ADT for metastatic disease, OR
o > 1 year since prior ADT for early-stage disease with good performance status
• Patients may receive only one of these agents (apalutamide, enzalutamide, or
abiraterone) in this setting and switching only if intolerant (without progression)
Alberta Health
Alendronate Osteoporosis 70 mg PO Multiple Regular Benefit: No form required Drug Benefit List
Generic
[3-22]
Group 2* AHS Outpatient
Cancer Drug
Eligibility: Benefit Program
• Apalutamide in combination with ADT for the treatment of patients with nmCRPC who are [3-22]
Apalutamide Not at high risk of developing metastases. High risk defined as:
(Erleada) nmCRPC Oral specified o PSADT ≤ 10 months, during continuous ADT/post orchiectomy
Janssen
• Patients may receive only one of these agents (darolutamide, apalutamide or
enzalutamide) in this setting and switching only if intolerant (without progression)
Page 2 | © Canadian Urological Association
v.01-MAR-2022