Page 10 - Flipbook
P. 10
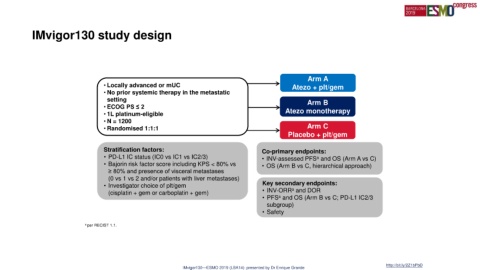
IMvigor130 study design
Arm A
• Locally advanced or mUC Atezo + plt/gem
• No prior systemic therapy in the metastatic
setting Arm B
• ECOG PS ≤ 2
• 1L platinum-eligible Atezo monotherapy
• N = 1200
• Randomised 1:1:1 Arm C
Placebo + plt/gem
Stratification factors: Co-primary endpoints:
• PD-L1 IC status (IC0 vs IC1 vs IC2/3) • INV-assessed PFS and OS (Arm A vs C)
a
• Bajorin risk factor score including KPS < 80% vs • OS (Arm B vs C, hierarchical approach)
≥ 80% and presence of visceral metastases
(0 vs 1 vs 2 and/or patients with liver metastases)
• Investigator choice of plt/gem Key secondary endpoints:
a
(cisplatin + gem or carboplatin + gem) • INV-ORR and DOR
• PFS and OS (Arm B vs C; PD-L1 IC2/3
a
subgroup)
• Safety
a per RECIST 1.1.
http://bit.ly/2Z1bPbD
IMvigor130—ESMO 2019 (LBA14): presented by Dr Enrique Grande