Page 5 - CUA Adv Prostate Ca Drug Acccess Listing
P. 5
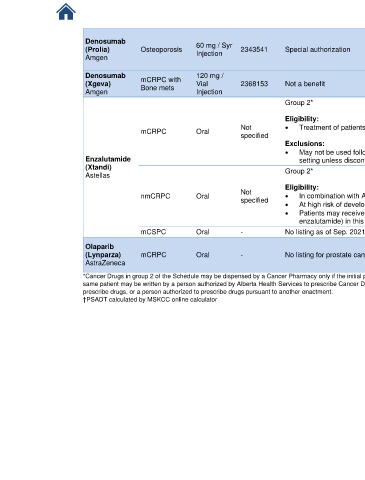
AB BlueCross
Denosumab Drug Benefit List
(Prolia) Osteoporosis 60 mg / Syr 2343541 Special authorization
Injection
Amgen
Denosumab mCRPC with 120 mg / AB BlueCross
(Xgeva) Vial 2368153 Not a benefit Drug Benefit List
Amgen Bone mets Injection
Group 2* AB Cancer
Outpatient Drug
Eligibility: Benefit Program
Not • Treatment of patients with mCRPC [9-21]
mCRPC Oral
specified
Exclusions:
• May not be used following apalutamide, enzalutamide, or darolutamide use in nmCRPC
Enzalutamide setting unless discontinuation due to intolerance (without progression)
(Xtandi) Group 2* AB Cancer
Astellas
Outpatient Drug
Eligibility: Benefit Program
nmCRPC Oral Not • In combination with ADT for the treatment of patients with nmCRPC [9-21]
specified
• At high risk of developing metastases (PSADT ≤ 10 months during continuous ADT)
†
• Patients may receive only one of these agents (darolutamide, apalutamide or
enzalutamide) in this setting and switching only if intolerance, not progression
mCSPC Oral - No listing as of Sep. 2021
AB Cancer
Olaparib Outpatient Drug
(Lynparza) mCRPC Oral - No listing for prostate cancer as of Sep. 2021 Benefit Program
AstraZeneca [9-21]
*Cancer Drugs in group 2 of the Schedule may be dispensed by a Cancer Pharmacy only if the initial prescription is written by a Cancer Centre Medical Staff member, but a subsequent prescription for the
same patient may be written by a person authorized by Alberta Health Services to prescribe Cancer Drugs and who is a physician, a regulated member under the Health Professions Act authorized to
prescribe drugs, or a person authorized to prescribe drugs pursuant to another enactment.
†PSADT calculated by MSKCC online calculator
Page 3 | © Canadian Urological Association
v.01-SEP-2021