Page 23 - Flipbook
P. 23
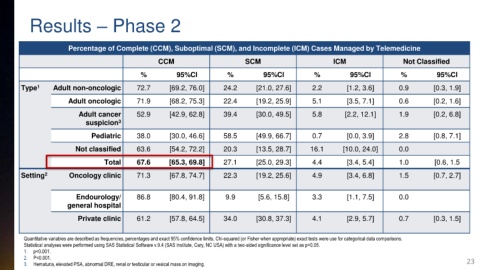
Results – Phase 2
Percentage of Complete (CCM), Suboptimal (SCM), and Incomplete (ICM) Cases Managed by Telemedicine
CCM SCM ICM Not Classified
% 95%CI % 95%CI % 95%CI % 95%CI
Type 1 Adult non-oncologic 72.7 [69.2, 76.0] 24.2 [21.0, 27.6] 2.2 [1.2, 3.6] 0.9 [0.3, 1.9]
Adult oncologic 71.9 [68.2, 75.3] 22.4 [19.2, 25.9] 5.1 [3.5, 7.1] 0.6 [0.2, 1.6]
Adult cancer 52.9 [42.9, 62.8] 39.4 [30.0, 49.5] 5.8 [2.2, 12.1] 1.9 [0.2, 6.8]
suspicion 3
Pediatric 38.0 [30.0, 46.6] 58.5 [49.9, 66.7] 0.7 [0.0, 3.9] 2.8 [0.8, 7.1]
Not classified 63.6 [54.2, 72.2] 20.3 [13.5, 28.7] 16.1 [10.0, 24.0] 0.0
Total 67.6 [65.3, 69.8] 27.1 [25.0, 29.3] 4.4 [3.4, 5.4] 1.0 [0.6, 1.5
Setting 2 Oncology clinic 71.3 [67.8, 74.7] 22.3 [19.2, 25.6] 4.9 [3.4, 6.8] 1.5 [0.7, 2.7]
Endourology/ 86.8 [80.4, 91.8] 9.9 [5.6, 15.8] 3.3 [1.1, 7.5] 0.0
general hospital
Private clinic 61.2 [57.8, 64.5] 34.0 [30.8, 37.3] 4.1 [2.9, 5.7] 0.7 [0.3, 1.5]
Quantitative variables are described as frequencies, percentages and exact 95% confidence limits. Chi-squared (or Fisher when appropriate) exact tests were use for categorical data comparisons.
Statistical analyses were performed using SAS Statistical Software v.9.4 (SAS Institute, Cary, NC USA) with a two-sided significance level set as p<0.05.
1. p<0.001.
2. P<0.001. 23
3. Hematuria, elevated PSA, abnormal DRE, renal or testicular or vesical mass on imaging.